Table of Contents
Introduction: Walk-In Interview at RPG Life Sciences Limited – Production, Packing, QA, QC & Engineering Departments
Welcome to MoneyJobs.in – We are excited to inform you about Walk-In Interview at RPG Life Sciences Limited – Production, Packing, QA, QC & Engineering Departments.
Don’t miss this golden opportunity to join one of the leading names in the Chemical industry. Stay updated with detailed job descriptions, eligibility criteria, and interview schedules only on MoneyJobs.in – your next career move starts here!
About RPG Life Sciences Limited
RPG Life Sciences Limited is a well-established pharmaceutical company in India, known for its high-quality formulations and strong regulatory compliance standards. The company operates advanced manufacturing facilities that adhere to GMP norms and cater to both domestic and international markets. With a focus on innovation, quality assurance, and operational excellence, RPG Life Sciences has built a solid reputation in the pharmaceutical manufacturing sector.
The walk-in interview is being conducted for its Formulation Manufacturing Plant located at Ankleshwar, one of Gujarat’s major industrial hubs under Gujarat Industrial Development Corporation (GIDC).
Manufacturing Units:
- Formulation Manufacturing Plant (OSD – Oral Solid Dosage)
- Quality Control & Quality Assurance Units
- Engineering & Instrumentation Department
- Packing & Liquid Manufacturing Section
Manufactures:
- Oral Solid Dosage (OSD) formulations
- Tablets, Capsules
- Liquid formulations
- Blister packaging and finished dosage forms
Website:-
https://www.rpglifesciences.com
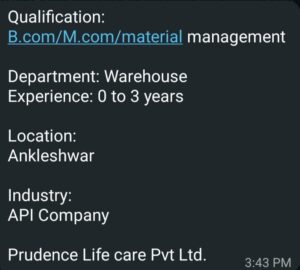
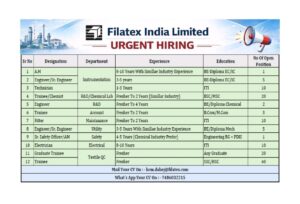
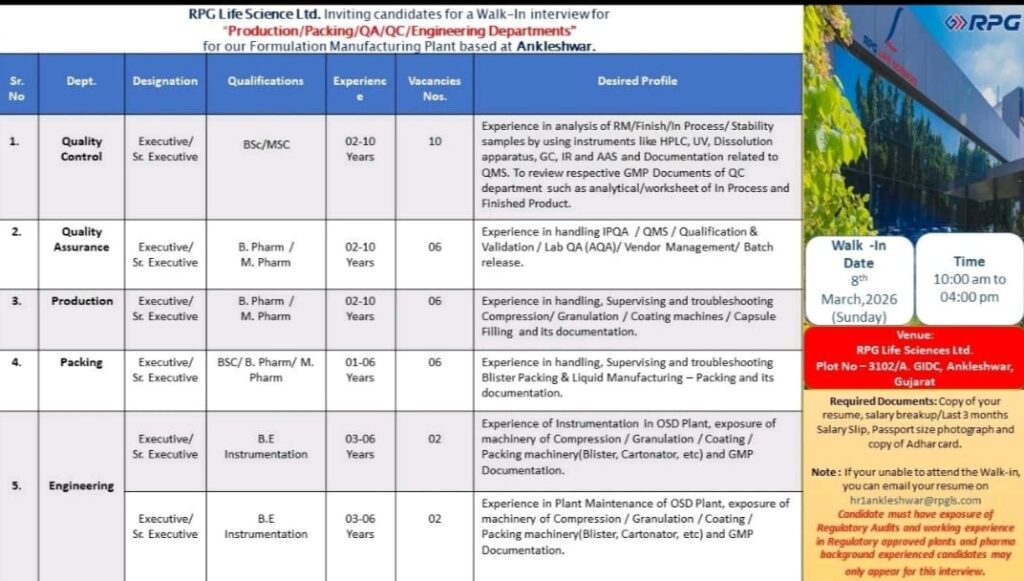
Open Positions and Job Details: RPG Life Sciences Limited
1. Position: Executive / Sr. Executive
Department: Quality Control
Total Requirement: 10
Qualification: B.Sc / M.Sc
Years of Experience: 02 – 10 Years
Responsibilities:
- Analysis of Raw Materials (RM), Finished Products, In-Process samples, and Stability samples
- Handling and operation of analytical instruments such as HPLC, UV, Dissolution Apparatus, GC, IR, and AAS
- Documentation related to QMS
- Review of GMP documents related to QC department
- Preparation and review of analytical worksheets for in-process and finished products
2. Position: Executive / Sr. Executive
Department: Quality Assurance
Total Requirement: 06
Qualification: B. Pharm / M. Pharm
Years of Experience: 02 – 10 Years
Responsibilities:
- Handling IPQA activities
- QMS documentation and compliance
- Qualification & Validation processes
- Lab QA (AQA) activities
- Vendor management
- Batch release documentation and review
3. Position: Executive / Sr. Executive
Department: Production
Total Requirement: 06
Qualification: B. Pharm / M. Pharm
Years of Experience: 02 – 10 Years
Responsibilities:
- Handling, supervising, and troubleshooting Compression, Granulation, and Coating machines
- Capsule filling operations
- Ensuring production documentation compliance
- Maintaining GMP standards in manufacturing operations
4. Position: Executive / Sr. Executive
Department: Packing
Total Requirement: 06
Qualification: B.Sc / B. Pharm / M. Pharm
Years of Experience: 01 – 06 Years
Responsibilities:
- Handling and supervising Blister Packing operations
- Experience in Liquid Manufacturing – Packing
- Troubleshooting packing machinery
- Maintaining proper documentation and GMP compliance
5. Position: Executive / Sr. Executive
Department: Engineering (Instrumentation)
Total Requirement: 02
Qualification: B.E. Instrumentation
Years of Experience: 03 – 06 Years
Responsibilities:
- Instrumentation experience in OSD plant
- Exposure to Compression, Granulation, Coating, Packing machinery, Blister & Cartonator systems
- Ensuring GMP documentation and compliance
- Preventive maintenance and troubleshooting
6. Position: Executive / Sr. Executive
Department: Engineering (Plant Maintenance)
Total Requirement: 02
Qualification: B.E. Instrumentation
Years of Experience: 03 – 06 Years
Responsibilities:
Handling breakdown maintenance and preventive maintenance schedules
Plant maintenance of OSD manufacturing unit
Exposure to Compression, Granulation, Coating, Packing machinery, Blister & Cartonator systems
Ensuring compliance with GMP documentation
Job Location: RPG Life Sciences Limited
Formulation Manufacturing Plant, Plot No. 3102/A, GIDC, Ankleshwar, Gujarat.
How to Apply: RPG Life Sciences Limited
Walk-In Interview Details:
📅 Date: 8th March 2026 (Sunday)
⏰ Time: 10:00 AM to 04:00 PM
📍 Venue: RPG Life Sciences Ltd., Plot No. 3102/A, GIDC, Ankleshwar
Required Documents:
- Updated Resume
- Copy of latest salary breakup
- Last 3 months’ salary slips
- Passport size photograph
- Copy of Aadhaar Card
If candidates are unable to attend the walk-in interview, they may email their updated resume to the official HR email mentioned in the advertisement.
Important Note:
Candidates must have exposure to regulatory audits and experience in regulatory-approved pharmaceutical plants. Preference will be given to candidates with prior pharma background and audit exposure.
Final Words: Walk-In Interview at RPG Life Sciences Limited – Production, Packing, QA, QC & Engineering Departments
This is an excellent opportunity for pharma professionals seeking career growth in Quality Control, Quality Assurance, Production, Packing, and Engineering domains. RPG Life Sciences offers a structured work environment, strong regulatory compliance culture, and opportunities to work with advanced pharmaceutical manufacturing technologies.
Interested and eligible candidates are encouraged to attend the walk-in interview with all required documents. Take the next step in your pharmaceutical career with a reputed and growing organization.